
Japan Diagnostic Labs Market Research Report: Forecast (2026-2032)
Japan Diagnostic Labs Market - By Test Type (Pathology, Radiology & Imaging), By Pathology Test Type (Clinical Chemistry, Hematology, Immunology & Serology, Microbiology, Histopath ... ology & Cytopathology, Molecular Diagnostics, Genetic Testing), By Radiology Type (X-Ray, Ultrasound, CT Scan, MRI, Mammography, PET-CT, Nuclear Imaging, Others), By Service Delivery Mode (Walk-in Testing, Home Sample Collection, Mobile Diagnostic Units), By Disease Type (Infectious Diseases, Oncology, Diabetes & Endocrinology, Cardiology, Neurology, Nephrology, Gastroenterology, Gynecology & Obstetrics, Respiratory Disorders, Orthopedics, Others), By End-User (Hospitals, Physician Clinics, Independent Diagnostic Centers, Corporate & Preventive Health Programs, Government & Public Healthcare Institutions), and others Read more
- Healthcare
- Feb 2026
- Pages 138
- Report Format: PDF, Excel, PPT
Japan Diagnostic Labs Market
Projected 5.08% CAGR from 2026 to 2032
Study Period
2026-2032
Market Size (2025)
USD 1.26 Billion
Market Size (2032)
USD 2.83 Billion
Base Year
2025
Projected CAGR
5.08%
Leading Segments
By Disease Type: Oncology
Japan Diagnostic Labs Market Report Key Takeaways:
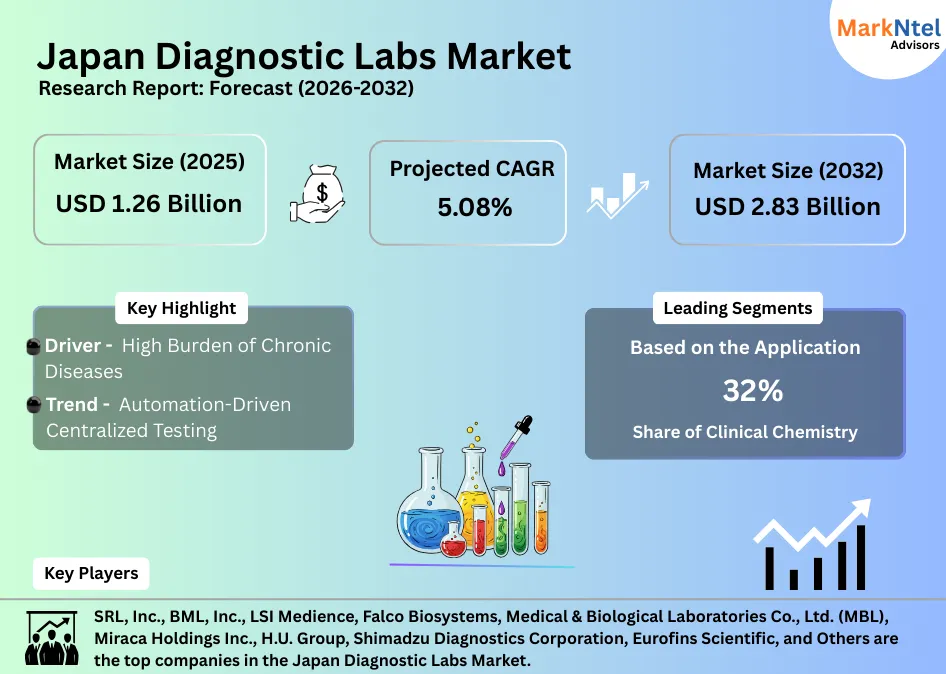
- Market size was valued at around USD1.26 billion in 2025 and is projected to reach USD2.83 billion by 2032. The estimated CAGR from 2026 to 2032 is around 5.08%, indicating strong growth.
- By Pathology test type, the Clinical Chemistry segment represented a significant share of about 32% in the Japan Diagnostic Labs Market in 2025.
- By Disease Type, the Oncology segment presented a significant share of about 28% in the Japan Diagnostic Labs Market in 2025.
- Leading Diagnostic Lab Companies in the Japan Market are SRL, Inc., BML, Inc., LSI Medience, Falco Biosystems, Medical & Biological Laboratories Co., Ltd. (MBL), Miraca Holdings Inc., H.U. Group, Shimadzu Diagnostics Corporation, Eurofins Scientific, and Others.
Market Insights & Analysis: Japan Diagnostic Labs Market (2026-32):
The Japan Diagnostic Labs Market size was valued at around USD1.26 billion in 2025 and is projected to reach USD2.83 billion by 2032. Along with this, the market is estimated to grow at a CAGR of around 5.08% during the forecast period, i.e., 2026-32.
Japan’s Diagnostic ecosystem is among the most utilization-intensive globally, supported by universal health coverage, mandatory health checkups, and high outpatient engagement. According to the Ministry of Health, Labor and Welfare, Japan records over 1.7 billion outpatient visits annually , directly translating into sustained laboratory test volumes across clinical chemistry, hematology, and immunology.
Additionally, market dynamics are shaped by hospital-centric diagnostics and large reference laboratory networks. Japan has around 3,200 general hospitals, most operating in-house laboratories for routine testing, while outsourcing high-complexity molecular and genetic assays to centralized labs. Diagnostic utilization is structurally high, as Japan averages more than 12 physician consultations per capita per year , significantly above OECD averages, reinforcing continuous laboratory demand.
Investment activity is concentrated in automation and centralization rather than greenfield expansion. Leading diagnostic groups have invested heavily in mega-scale central laboratories equipped with total laboratory automation, robotics, and AI-enabled analyzers to process millions of samples annually. For instance, major lab operators expanded high-throughput central testing facilities and nationwide sample logistics networks to improve turnaround times and mitigate workforce shortages. Digital pathology platforms and laboratory information system upgrades have also been deployed to support remote reporting and workflow efficiency.
The market is further supported by aging demographics and oncology-led testing intensity. According to the Statistics Bureau of Japan, 29.1% of the population was aged more than 65 in 2024 , driving repeated diagnostic monitoring. Combined with rising cancer prevalence, expanded molecular diagnostics, and precision medicine adoption, these structural and investment-led factors sustain strong growth momentum through 2032.
Japan Diagnostic Labs Market Recent Developments:
- 2025 : In 2025, Fujirebio (H.U. Group) secured regulatory expansion of its Lumipulse® G Beta-Amyloid assays for Alzheimer’s diagnostics, increasing hospital lab adoption.
Japan Diagnostic Labs Market Scope:
| Category | Segments |
|---|---|
| By Test Type | Pathology, Radiology & Imaging |
| By Pathology Test Type | Clinical Chemistry, Hematology, Immunology & Serology, Microbiology, Histopathology & Cytopathology, Molecular Diagnostics, Genetic Testing |
| By Radiology Type | X-Ray, Ultrasound, CT Scan, MRI, Mammography, PET-CT, Nuclear Imaging, Others |
| By Service Delivery Mode | Walk-in Testing, Home Sample Collection, Mobile Diagnostic Units |
| By Disease Type | Infectious Diseases, Oncology, Diabetes & Endocrinology, Cardiology, Neurology, Nephrology, Gastroenterology, Gynecology & Obstetrics, Respiratory Disorders, Orthopedics, Others |
| By End-User | Hospitals, Physician Clinics, Independent Diagnostic Centers, Corporate & Preventive Health Programs, Government & Public Healthcare Institutions |
Japan Diagnostic Labs Market Driver:
High Burden of Chronic Diseases
The growing prevalence of chronic non-communicable diseases is a major driver of Japan’s diagnostic labs market, as these conditions require continuous laboratory monitoring over long treatment durations. Unlike acute illnesses, chronic diseases generate recurring demand for routine and specialized diagnostic tests, anchoring stable test volumes for diagnostic laboratories.
Cardiovascular and metabolic disorders account for a substantial share of diagnostic utilization. According to the Ministry of Health, Labor and Welfare (MHLW) Patient Survey 2023, approximately 16.09 million people were receiving treatment for hypertension, 5.52 million for diabetes, and 4.01 million for lipid metabolism disorders . Management of these conditions depends on repeated clinical chemistry tests, glucose and HbA1c monitoring, lipid panels, electrolyte analysis, and renal function testing, driving high-frequency laboratory workloads throughout the year.
Neurological, renal, and respiratory diseases further intensify diagnostic demand. MHLW data show that 1.88 million patients were treated for cerebrovascular diseases, 0.63 million for chronic kidney disease, and 0.38 million for chronic obstructive pulmonary disease in 2023. These conditions require ongoing laboratory surveillance, including coagulation studies, kidney function panels, inflammatory markers, and blood gas analysis.
Japan Diagnostic Labs Market Trend:
Automation-Driven Centralized Testing
Automation-driven centralization is strengthening in Japan as new laboratory platforms combine robotics, digital connectivity, and high-throughput design, allowing testing to shift from small hospital labs to large automated core facilities. For instance, Roche Diagnostics, in 2025, launched 61 new diagnostic products, including 2 new core platforms and 6 digital workflow solutions , designed to integrate instruments, data flow, and result reporting across laboratory networks.
These systems enable centralized labs to process very high sample volumes while digitally serving multiple hospitals and clinics from one site. Additionally, Roche’s cobas® analytical systems, capable of handling up to 2,000 tests per hour , directly support consolidation by replacing multiple low-volume labs with a single automated hub.
Likewise, Japanese manufacturer Hitachi High-Tech expanded its LabFLEX automation systems in Japan, which automate pre-analytical steps such as tube sorting, decapping, aliquoting, and transport. These steps are critical for central labs receiving specimens from many external locations, as automation ensures consistent handling and traceability at scale.
Japan Diagnostic Labs Market Opportunity:
Rising Investments in Healthcare Infrastructure
The Japanese government’s investments in healthcare R&D, genomic data platforms, and medical device innovation are strengthening the diagnostic labs market by creating funding, infrastructure, and data ecosystems that labs can participate in. The Japan Agency for Medical Research and Development (AMED), funded by the Cabinet Secretariat, Ministry of Health, Labor and Welfare (MHLW), Ministry of Education, Culture, Sports, Science and Technology (MEXT), and Ministry of Economy, Trade and Industry (METI), has 2025 budget allocation of about USD1.0 billion for research and development in medical technologies, health data, and diagnostics . This centralized agency supports projects spanning medical devices, genomic research, and health data platforms, enabling labs to engage in funded R&D collaborations and advanced diagnostic development.
Government grant programs under AMED, such as AMED-CREST and PRIME advanced research funds, prioritize breakthrough technologies, including next-generation diagnostics, measurement systems, and genome-based tools. These programs also include long-term, multi-year funding and public–private matching grants that connect clinical research institutions with industry partners.
In 2025, Japan’s MHLW also increased its overall health budget by more than USD3 billion , with portions allocated to strengthening regulatory capacity, medical device evaluation systems, and innovation support, improving the approval pipeline and commercialization environment for advanced diagnostic instruments.
Japan Diagnostic Labs Market Challenge:
Shortage of Skilled Workforce Impeding Market Growth
Japan’s healthcare system is facing a structural labor shortfall driven by rapid population aging and a shrinking working-age population. For instance, the Cabinet Secretariat of Japan projects a shortage of around 300,000 healthcare workers by 2040 , even after accounting for productivity improvements. This shortage directly affects medical technologists, clinical laboratory technicians, and pathology staff, who are essential for diagnostic lab operations. Likewise, advanced testing areas such as genomic diagnostics and molecular pathology intensify the problem because they require highly trained personnel, longer training cycles, and compliance with strict quality standards. As genetic testing volumes rise under national reimbursement schemes, workforce constraints increasingly limit scalability.
Together, demographic decline, uneven geographic distribution of professionals, and rising test complexity make workforce shortages a core operational and cost challenge for Japan’s diagnostic laboratories, accelerating pressure to automate, centralize, and outsource testing.
Japan Diagnostic Labs Market (2026-32) Segmentation Analysis:
The Japan Diagnostic Labs Market study of MarkNtel Advisors evaluates & highlights the major trends and influencing factors in each segment. It includes predictions for the period 2026–32 at the country level. Based on the analysis, the market has been further classified as;
Based on Pathology Test Type:
- Clinical Chemistry
- Hematology
- Immunology & Serology
- Microbiology
- Histopathology & Cytopathology
- Molecular Diagnostics
- Genetic Testing
Clinical chemistry is the largest and most consistent volume driver in this market with a market share of about 32%, because it is tightly linked to everyday clinical decision-making, long-term disease monitoring, and preventive healthcare. Routine biochemistry panels, such as glucose, HbA1c, lipid profiles, liver enzymes, kidney function markers, and electrolytes, are ordered repeatedly for the same patients over time, unlike one-time or episodic tests. This makes chemistry testing continuous rather than occasional.
Additionally, Japan’s lab ecosystem is structurally aligned with chemistry testing. Around 66,000 licensed clinical laboratory technologists form the core workforce , and their education and routine workflows are historically centered on automated biochemistry analyzers. This workforce readiness, combined with widespread automation and standardized methods, allows chemistry testing to scale efficiently. Together, repeat clinical demand, chronic disease management, workforce specialization, and automation make clinical chemistry the most dominant and operationally essential segment in Japan’s diagnostic labs market.
Based on Disease Type:
- Infectious Diseases
- Oncology
- Diabetes & Endocrinology
- Cardiology
- Neurology
- Nephrology
- Gastroenterology
- Gynecology & Obstetrics
- Respiratory Disorders
- Orthopedics
- Others
Oncology dominates this market with a market share of about 28% because disease burden, government policy, reimbursement, and data infrastructure are all aligned around cancer care. Cancer has been Japan’s leading cause of death for over 40 years, accounting for about thousands of deaths annually, which forces continuous demand for diagnostics across the entire care cycle, screening, diagnosis, staging, therapy selection, and recurrence monitoring. This clinical burden feeds directly into laboratories through mandatory national systems.
For instance, the National Cancer Center Japan operates the National Cancer Registry, which legally requires hospitals to submit standardized cancer diagnostic data. This obligation structurally locks in demand for histopathology, cytology, immunohistochemistry, and tumor marker testing across Japan’s hospitals.
Gain a Competitive Edge with Our Japan Diagnostic Labs Market Report:
- Japan Diagnostic Labs Market Report by MarkNtel Advisors provides a detailed & thorough analysis of market size & share, growth rate, competitive landscape, and key players. This comprehensive analysis helps businesses gain a holistic understanding of the market dynamics & make informed decisions.
- This report also highlights current market trends & future projections, allowing businesses to identify emerging opportunities & potential challenges. By understanding market forecasts, companies can align their strategies & stay ahead of the competition.
- Japan Diagnostic Labs Market Report aids in assessing & mitigating risks associated with entering or operating in the market. By understanding market dynamics, regulatory frameworks, and potential challenges, businesses can develop strategies to minimize risks & optimize their operations.
*Reports Delivery Format - Market research studies from MarkNtel Advisors are offered in PDF, Excel and PowerPoint formats. Within 24 hours of the payment being successfully received, the report will be sent to your email address.
Frequently Asked Questions
- Market Segmentation
- Introduction
- Product Definition
- Research Process
- Assumptions
- Executive Summary
- Japan Diagnostic Labs Market Policies, Regulations, and Product Standards
- Japan Diagnostic Labs Market Trends & Developments
- Japan Diagnostic Labs Market Dynamics
- Growth Factors
- Challenges
- Japan Diagnostic Labs Market Hotspot & Opportunities
- Japan Diagnostic Labs Market Outlook, 2022-2032F
- Market Size & Outlook
- By Revenues (USD Million)
- Market Share & Outlook
- By Test Type- Market Size & Forecast 2022-2032, USD Million
- Pathology
- Radiology & Imaging
- By Pathology Test Type- Market Size & Forecast 2022-2032, USD Million
- Clinical Chemistry
- Hematology
- Immunology & Serology
- Microbiology
- Histopathology & Cytopathology
- Molecular Diagnostics
- Genetic Testing
- By Radiology Type- Market Size & Forecast 2022-2032, USD Million
- X-Ray
- Ultrasound
- CT Scan
- MRI
- Mammography
- PET-CT
- Nuclear Imaging
- Others
- By Service Delivery Mode- Market Size & Forecast 2022-2032, USD Million
- Walk-in Testing
- Home Sample Collection
- Mobile Diagnostic Units
- By Disease Type - Market Size & Forecast 2022-2032, USD Million
- Infectious Diseases
- Oncology
- Diabetes & Endocrinology
- Cardiology
- Neurology
- Nephrology
- Gastroenterology
- Gynecology & Obstetrics
- Respiratory Disorders
- Orthopedics
- Others
- By End-User- Market Size & Forecast 2022-2032, USD Million
- Hospitals
- Physician Clinics
- Independent Diagnostic Centers
- Corporate & Preventive Health Programs
- Government & Public Healthcare Institutions
- By Region- Market Size & Forecast 2022-2032, USD Million
- Greater Kanto
- Kansai
- Chubu
- Kyushu
- Hokkaido
- By Company
- Competition Characteristics
- Market Share & Analysis
- By Test Type- Market Size & Forecast 2022-2032, USD Million
- Market Size & Outlook
- Japan Pathology Labs Market Outlook, 2022-2032
- Market Size & Outlook
- By Revenues (USD Million)
- Market Share & Outlook
- By Pathology Test Type- Market Size & Forecast 2022-2032, USD Million
- By Radiology Type- Market Size & Forecast 2022-2032, USD Million
- By Service Delivery Mode- Market Size & Forecast 2022-2032, USD Million
- By Disease Type - Market Size & Forecast 2022-2032, USD Million
- By End-User- Market Size & Forecast 2022-2032, USD Million
- By Region- Market Size & Forecast 2022-2032, USD Million
- Market Size & Outlook
- Japan Radiology & Imaging Labs Market Outlook, 2022-2032
- Market Size & Outlook
- By Revenues (USD Million)
- Market Share & Outlook
- By Pathology Test Type- Market Size & Forecast 2022-2032, USD Million
- By Radiology Type- Market Size & Forecast 2022-2032, USD Million
- By Service Delivery Mode- Market Size & Forecast 2022-2032, USD Million
- By Disease Type - Market Size & Forecast 2022-2032, USD Million
- By End-User- Market Size & Forecast 2022-2032, USD Million
- By Region- Market Size & Forecast 2022-2032, USD Million
- Market Size & Outlook
- Japan Diagnostic Labs Market Key Strategic Imperatives for Success & Growth
- Competitive Outlook
- Company Profiles
- SRL, Inc.
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- BML, Inc.
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LSI Medience
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Falco Biosystems
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Medical & Biological Laboratories Co., Ltd. (MBL)
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Miraca Holdings Inc.
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- H.U. Group
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Shimadzu Diagnostics Corporation
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Eurofins Scientific
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Others
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- SRL, Inc.
- Company Profiles
- Disclaimer
MarkNtel Advisors follows a robust and iterative research methodology designed to ensure maximum accuracy and minimize deviation in market estimates and forecasts. Our approach combines both bottom-up and top-down techniques to effectively segment and quantify various aspects of the market. A consistent feature across all our research reports is data triangulation, which examines the market from three distinct perspectives to validate findings. Key components of our research process include:
1. Scope & Research Design At the outset, MarkNtel Advisors define the research objectives and formulate pertinent questions. This phase involves determining the type of research—qualitative or quantitative—and designing a methodology that outlines data collection methods, target demographics, and analytical tools. They also establish timelines and budgets to ensure the research aligns with client goals.
2. Sample Selection and Data Collection In this stage, the firm identifies the target audience and determines the appropriate sample size to ensure representativeness. They employ various sampling methods, such as random or stratified sampling, based on the research objectives. Data collection is carried out using tools like surveys, interviews, and observations, ensuring the gathered data is reliable and relevant.
3. Data Analysis and Validation Once data is collected, MarkNtel Advisors undertake a rigorous analysis process. This includes cleaning the data to remove inconsistencies, employing statistical software for quantitative analysis, and thematic analysis for qualitative data. Validation steps are taken to ensure the accuracy and reliability of the findings, minimizing biases and errors.

4. Data Forecast and FinalizationThe final phase involves forecasting future market trends based on the analyzed data. MarkNtel Advisors utilize predictive modeling and time series analysis to anticipate market behaviors. The insights are then compiled into comprehensive reports, featuring visual aids like charts and graphs, and include strategic recommendations to inform client decision-making









